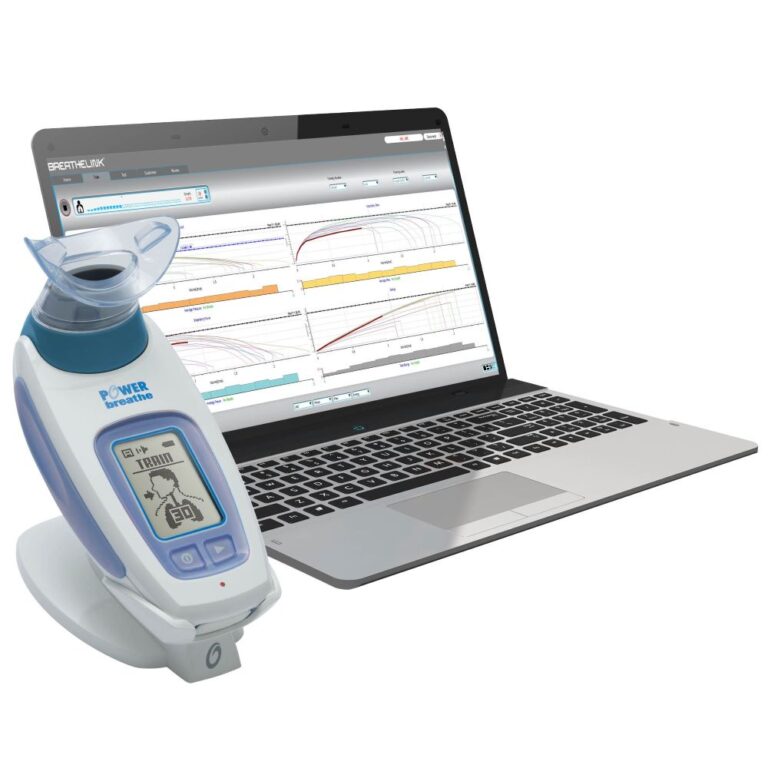
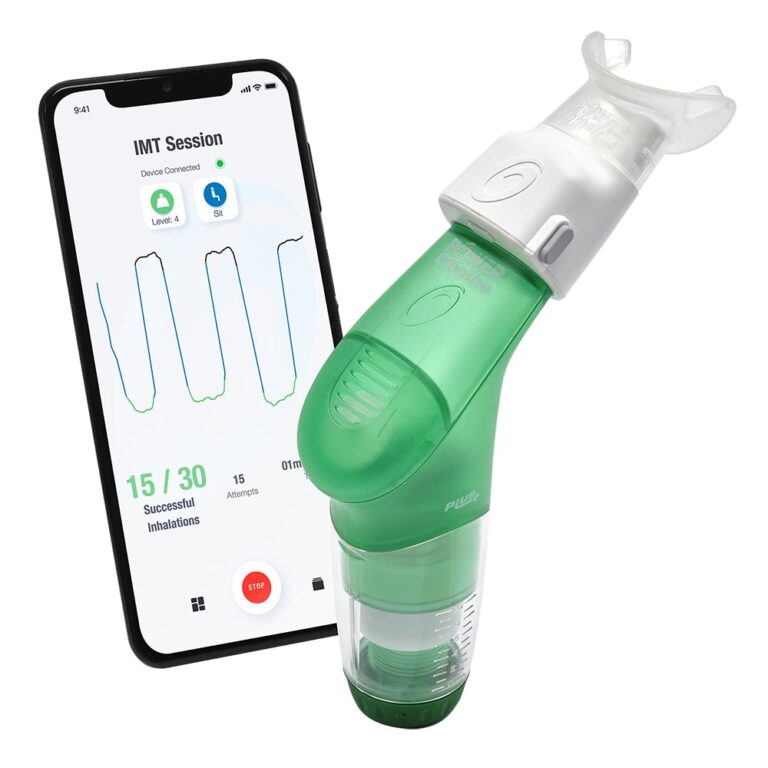
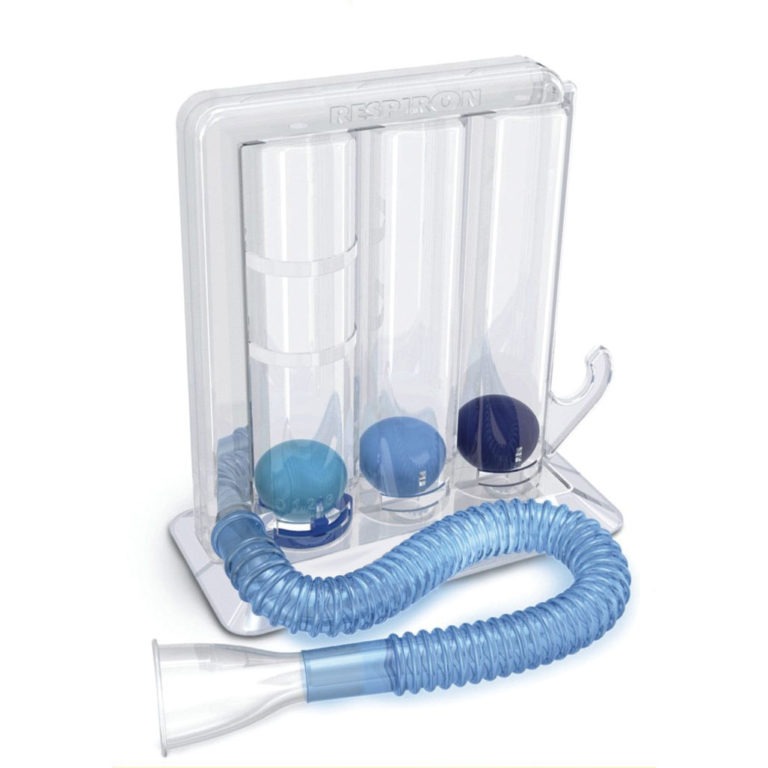
Breathing Trainers
The POWERbreathe range of drug-free breathing trainers include both Inspiratory Muscle Training (IMT & IMST) and Expiratory Muscle Training (EMT & EMST) devices.
-
£15.83